 
The bonds formed between the molecules always compete with excess water molecules in the surroundings, therefore because of this competition, the bonds formed are weaker when dissolved in water. 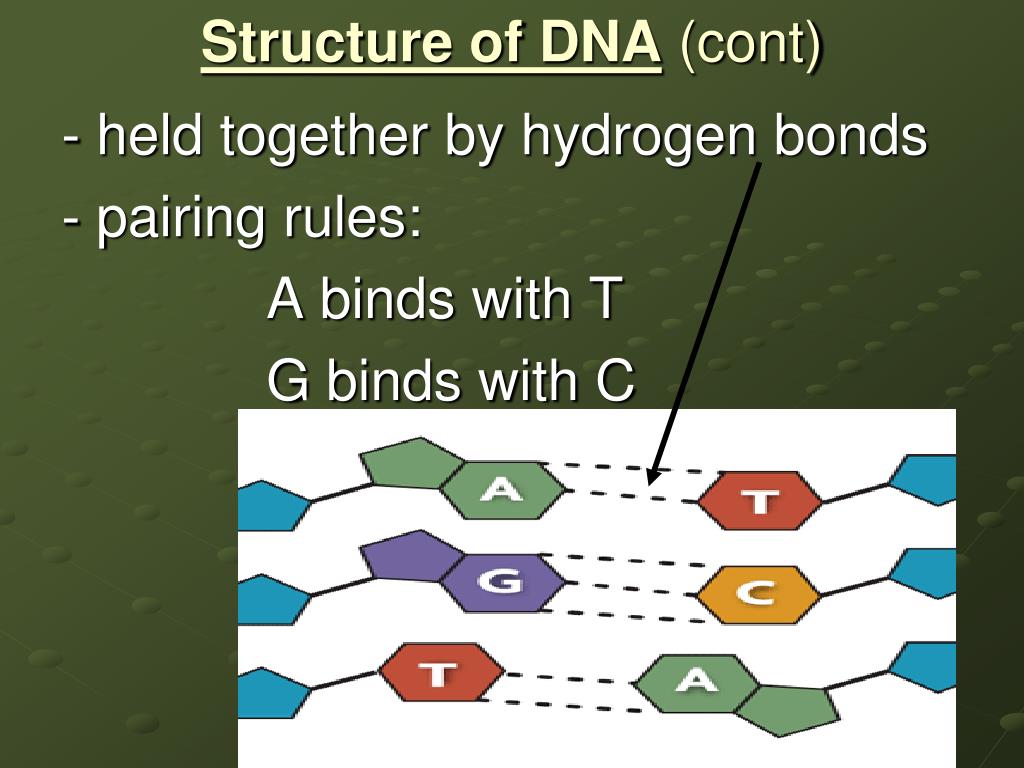
The bonds that are interior to the helix occur between the complementary base pairs.Įxterior bonds are formed between polar groups on the surface of nitrogen bases and surrounding water molecules. They form both internal and external hydrogen bonding in the DNA molecule. They constitute the strongest intermolecular attraction, but they are weaker than covalent and ionic interactions. Hydrogen bonds are usually formed between atoms of different molecule or in parts of the same molecule hence they are termed as intermolecular hydrogen bonding or intramolecular hydrogen bonding, respectively. The number of hydrogen bonds is directly proportional to their melting temperature, i.e, higher temperatures are required to disrupt the hydrogen bonds when they are many in number. The hydrogen bonding occurs between complementary purine and pyramidine bases. It is usually represented by a dotted line. Therefore, a strong non-covalent interaction occurs between hydrogen atom (donor) and electronegative atom (acceptor) with at least one lone pair of electrons forming a hydrogen bond. Hydrogen atoms attached to these electronegative groups act as hydrogen bond donors because of their strong partial positive charge. On DNA, since oxygen and nitrogen are highly electronegative atoms with free lone pair of electrons, they act as hydrogen bond acceptors. Hydrogen bonding is a dipole-dipole attraction between a hydrogen atom that is covalently bonded to a very high electronegative atom such as nitrogen, oxygen, and fluorine and other electronegative atoms.Įlectronegativity is defined as the tendency of an atom or molecule to accept electrons towards itself, resulting in the formation of dipoles and bonds. Crick in 20th century as “two sugar phosphate chains running anti-parallel to each other, paired with purine-pyramidine bases and are held together by hydrogen bonds.” The double helical structure of DNA was discovered by J.D.Watson and F.H.C. 
They also contribute to the double helical structure to the DNA. Overall, these bonds stabilize and maintain the structural integrity of the DNA by keeping the two strands intact. Usually, two hydrogen bonds are formed between adenine and thymine pairs, while three hydrogen bonds occur between cytosine and guanine pairs. They constitute the strongest intermolecular attractions but they weaker than covalent and ionic interactions.īoth interior and exterior types of hydrogen bonding are observed on the DNA. On DNA, since oxygen and nitrogen are highly electronegative atoms with free lone pair of electrons they act as hydrogen bond acceptors.īoth intermolecular and intramolecular hydrogen bonding exists on DNA. 
It is a dipole-dipole attraction between a hydrogen atom that is covalently bonded to a very high electronegative atom such as nitrogen, oxygen, and fluorine, and other electronegative atoms. Hydrogen bonding is a type of interaction that is formed between two electronegative atoms which are usually oxygen or nitrogen atoms in the nitrogenous bases of DNA. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
